Welcome to
Seattle Plastic Surgery Center
Today’s advanced cosmetic surgery techniques can make significant improvements in your appearance and self esteem. Choosing the right surgeon and having realistic expectations about the outcome are essential in the process of undergoing cosmetic surgery. Our board certified cosmetic and plastic surgeons offer comprehensive breast, body and face surgery procedures, artistically applied injectable products, and non-surgical alternatives custom tailored to meet each patient’s needs and desires.
Search by Procedure
Photo Gallery
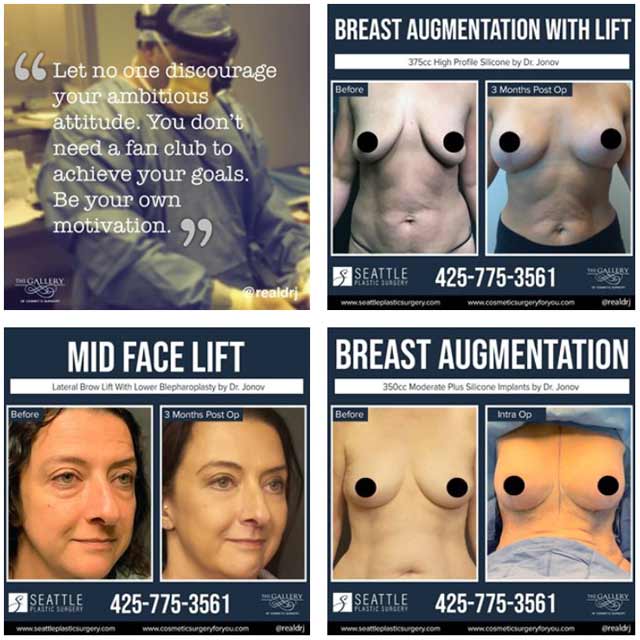
Before & After pictures
Browse through some of our patients’ amazing before and after pictures to get an idea of the aesthetic possibilities at Seattle Plastic Surgery
View GalleryView Gallery

Dr. Craig Jonov
Seattle surgeon Dr. Jonov is a board certified cosmetic and reconstructive surgeon. He was chosen as one of Seattle’s “Top Plastic Surgeons” in KING5 News’ Best of Western Washington contest. Seattle Plastic Surgery Center has excellent reviews as a result of amazing results and care for their patients. Exceptional care, excellent results.
Certifications & Affiliations
- Board Certified: American Board of Cosmetic Surgery
- Physician & Surgeon, State of Washington
- American Academy of Cosmetic Surgery
- American Society of Liposuction Surgery
- American Society of Cosmetic Breast Surgery
- American Medical Association

Dr. David Santos
Dr. Santos brings decades of expertise as a premier facial plastic surgeon and a pioneer in the use of cutting-edge technology in his field to Seattle Plastic Surgery. As a top plastic surgeon, he is frequently called upon to educate other surgeons about the latest developments in medical technology and facial plastic surgery throughout the United States and abroad. He is often found at the forefront of developing medical technologies and often one of the first plastic surgeons to utilize the newest technologies and protocols, including performing procedures using local anesthesia to minimize risks and downtime for his patients.
Certifications & Affiliations
- Board Certified In Otolaryngology
- Board Certified Facial Plastic and Reconstructive Surgery
- American College of Surgeons
- American Medical Association
- Academy of Facial Plastic and Reconstructive Surgery
- NW Academy of Otolaryngology
- Physician and Surgeon, State of Washington
- King County Medical Society


Darya Shmidt, ARNP
Aesthetic Medicine ARNP
Darya has an extensive background as an Advanced Registered Nurse Practitioner who began her career in women’s health. Five years ago, she became an aesthetic injector in Beverly Hills, where she began helping patients find their inner beauty and restoring confidence for every patient. In 2023, she discovered Seattle Plastic Surgery and joined our team as an Aesthetic Medicine Provider. She is passionate about challenging the narrative around cosmetic injections and loves sharing how medical aesthetics is integral to the healthcare system.
Tina PA-C
Weight Loss Provider
Tina was born and raised in Fort Worth, Texas, but left her home state to explore the mountains of Washington. Drawn to the aesthetic medicine business, Tina graduated from UT Southwestern Medical Center in Dallas as a certified Physician Assistant. Before she graduated, she knew he wanted to relocate to Seattle and join our company. Her love for helping patients grow and her incredible knowledge brought her to our team!

Monthly Specials
Injections + Esthetics + Skincare
See our newest monthly specials! Check back every month for updated specials, offerings, and new procedures.

Free Skin Consultation
Whether you want to look younger, address acne, or maintain your current skin, our master estheticians can create a customized treatment plan and product recommendations based on your specific goals, needs, and concerns during a free skin consultation.
Learn MoreLearn MoreLearn more about our services
Facial Surgery
Seattle Plastic Surgery provides many procedures and treatments to improve the shape and appearance of your face and skin.
Body Procedures
Our surgeons offer many affordable solutions to help you feel comfortable and confident with the look and feel of your body.
Breast Augmentation
We offer many cosmetic surgery procedures to increase breast size, rejuvenate with a breast lift, or reduce volume with a breast reduction.
Botox & Filler Training
hands-on training with real patients
Become a certified cosmetic injector with our skilled physician instructors.
Our clinic offers the best cosmetic injection training in Seattle. When you enroll in our course, you will work with some of the top cosmetic providers and surgeons with years of aesthetic medicine experience. Through our program, you can advance your career, create a stable income, or begin your practice.
Learn MoreLearn MoreScar Protocol
Post-Surgical Scar Treatment
All surgery patients are placed on Scar Protocol. Scar Protocol is a five-phase scar cream system that minimizes scars after surgery.
Learn MoreLearn More
Real Patient Reviews
Yelp | Aug 3, 2018
By: Micaela
Coming from the world of medical esthetics it was important having the best of the best and Dr J came highly recommend. I came in for a tummy tuck and Dr. J gave me results I couldn’t even dream of. My stomach is rock hard, flat and tiny!!! I’m 9 days post operation and I feel great!!! The doctor has been super nice the through the whole process since the beginning with my consult. The staff is friendly the office is clean and super cute.. Between his skill, bedside manner and professionalism I don’t know why anyone would go anywhere else!!!
RealSelf | Jan 27, 2018
By: Jess
I had a Mommy Makeover (tummy tuck, muscle repair, Lipo of the flanks, breast reduction and breast lift) done in October with Dr. Jonov and I have nothing but nice things to say about him and his staff. He is a very talented surgeon. My results are just phenomenal! My pain was also very minimal. I seriously can’t believe how easy my whole recovery has been. I’m so so beyond happy with how everything is looking and I’m only 10 weeks out. And to think it’s just going to get better from here. Amazing! Dr. Jonov has been such a pleasure to work with.
Healthgrades | Jun 19, 2018
By: Everette
Dr. Jonov is amazing! After being a stay at home mom with 4 kids, I felt was time to reward myself. I went in for Mommy Makeover consultation and it was a great!. I was impressed with his staff and his honesty when it came to discuss my result. He mentioned I had no breast sag and would not need a breast lift, although I would need a full tummy tuck to get rid some of my stretchmarks. I am now 4 months post from my Mommy Makeover and I’m in love with the new me!